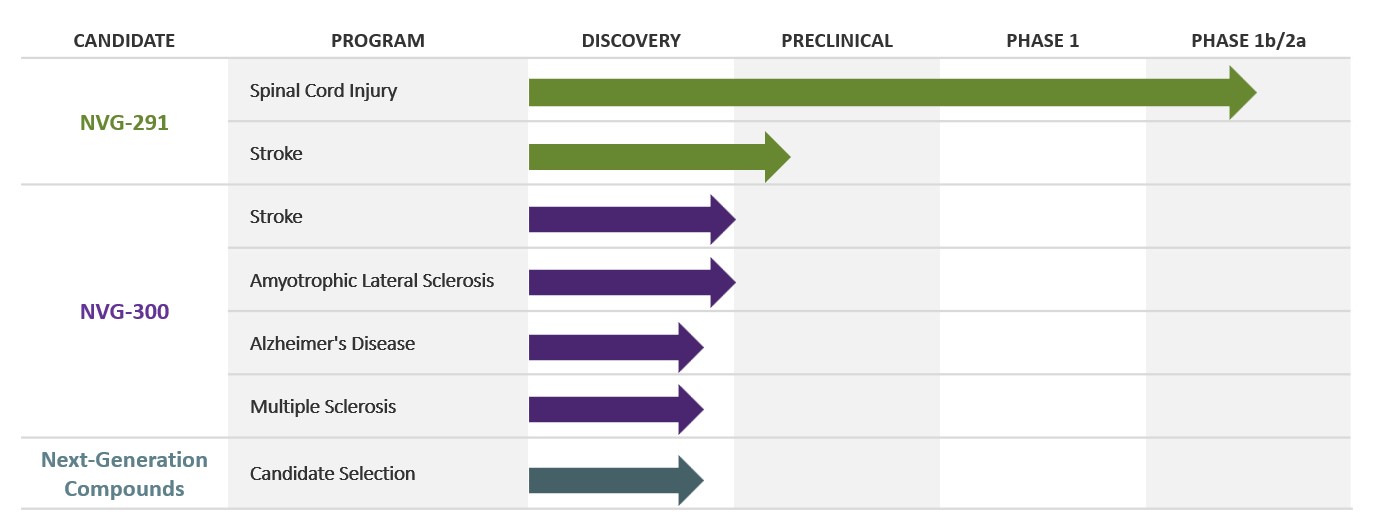
Pipeline
NervGen’s lead drug candidate, NVG-291, is a first-in-class therapeutic peptide targeting mechanisms that interfere with nervous system repair. NVG-291 is being evaluated in a Phase 1b/2a clinical trial. NervGen has initiated preclinical evaluation of a new development candidate, NVG-300, in models of ischemic stroke, amyotrophic lateral sclerosis (ALS) and spinal cord injury.
Nervous System Damage
When there is damage to the nervous system a scar forms. The scar forms in response to acute damage, such as spinal cord injury and traumatic brain injury, or in chronic diseases, such as Alzheimer’s and multiple sclerosis. Scars contain molecules called chondroitin sulfate proteoglycans (CSPGs), which initially help to contain damage, but in the long term interfere with repair of the nervous system. In animal studies, NVG-291-R promoted nervous system repair and enhanced recovery of functions such as walking, bladder control, vision, and memory.
We have completed our Phase 1 clinical trial and have initiated a Phase 1b/2a clinical trial to evaluate the efficacy of NVG-291 in individuals with either chronic (1-10 years post-injury) or subacute (those with a more recent injury) spinal cord injury.
We have also initiated preclinical evaluation of a new development candidate, NVG-300, in models of ischemic stroke, amyotrophic lateral sclerosis (ALS) and spinal cord injury.